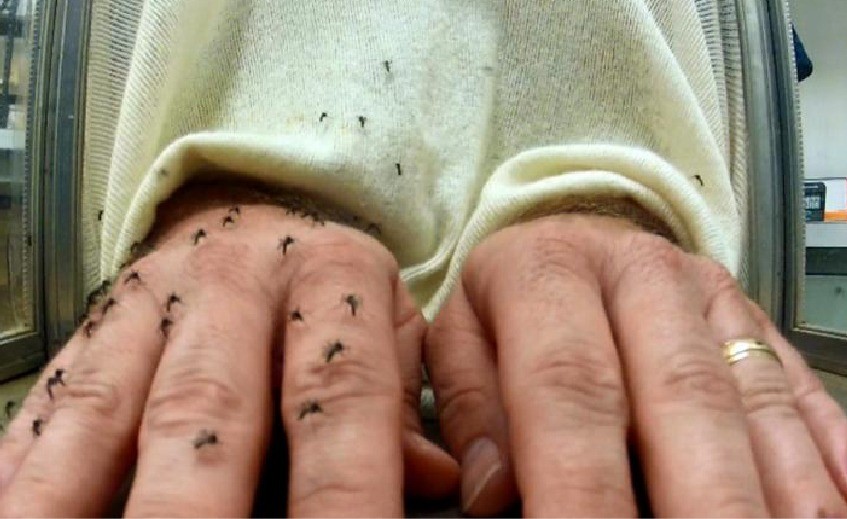
Procedure: The 018-A formulation of NO MAS was evaluated on skin for repellency to female Aedes aegypti mosquitoes. In a single repellent duration test, a 1 milliliter dose of the formulation was applied to the left forearm of the test volunteer. The formulation was spread evenly from wrist to elbow (on approximately 650 cm2 of skin surface area) and evaluated for repellency to caged female mosquitoes. One hour after the application of the formulation, the treated arm was exposed for 3 min to mosquitoes and then re-exposed in the same manner at hourly intervals thereafter until repellent failure. The test was conducted in a 40 cm3 cage containing 200 (6-9 day-old) female Aedes aegypti mosquitoes preselected for testing on the basis of demonstrated human host avidity. Repellency was calculated as Complete Protection Time (CPT), i.e., the time between repellent application and 2 or more probings of the skin on the treated arm, or the first probing followed by another probing within 30 min. A glove was worn to protect the untreated hand from mosquito bites during testing. Two negative controls were used in each test (observed once per hour until repellent failure) and comprised the number of female mosquitoes in the test cage and in a (separate) control cage that landed and remained on the untreated skin of the right forearm of the test volunteer for > 5 sec during a 10 second exposure period.